结核与肺部疾病杂志 ›› 2022, Vol. 3 ›› Issue (4): 328-333.doi: 10.19983/j.issn.2096-8493.20220073
收稿日期:2022-04-20
出版日期:2022-08-20
发布日期:2022-08-16
通信作者:
谢佳星
E-mail:jiaxingxie@126.com
基金资助:
Lin Huimin1, Fu Yu1, Fang Zhangfu1,2, Xie Jiaxing1,3( )
)
Received:2022-04-20
Online:2022-08-20
Published:2022-08-16
Contact:
Xie Jiaxing
E-mail:jiaxingxie@126.com
Supported by:摘要:
哮喘是临床常见的异质性慢性气道炎症性疾病,其中嗜酸性粒细胞哮喘(eosinophilic asthma,EA)是最常见的临床表型。持续性气道嗜酸性粒细胞炎症极易促使哮喘进展为重症,是导致哮喘高致残率及高致死率的原因之一。变应性EA与非变应性EA在临床特征、发病机制方面具有明显区别,深入认识EA发病机制对疾病的管理和改善患者预后具有重要意义。作者对EA的研究进展进行综述。
中图分类号:
林慧敏, 符昱, 方章福, 谢佳星. 嗜酸性粒细胞哮喘的研究进展[J]. 结核与肺部疾病杂志, 2022, 3(4): 328-333. doi: 10.19983/j.issn.2096-8493.20220073
Lin Huimin, Fu Yu, Fang Zhangfu, Xie Jiaxing. Research progress on eosinophilic asthma[J]. Journal of Tuberculosis and Lung Disease, 2022, 3(4): 328-333. doi: 10.19983/j.issn.2096-8493.20220073
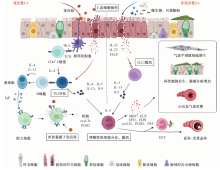
图1
变应性EA与非变应性EA发病机制 注 图中黑色虚线左侧为变应性嗜酸性粒细胞哮喘(eosinophilic asthma,EA)的免疫学机制通路,即变应原被树突状细胞(dendritic cell,DC)识别,并诱导DC分化,DC通过释放白细胞介素(interleukin,IL)-4诱导CD4+T细胞分化为2型辅助性T淋巴细胞(T helper lymphocytes 2,Th2);此外,变应原可通过刺激气道上皮细胞释放警报因子,如IL-33、IL-25及胸腺基质淋巴细胞生成素(thymic stromal lymphopoietin,TSLP),警报因子能够促进Th2细胞的活化。活化的Th2通过释放IL-4、IL-5、IL-13、IL-9等2型炎症因子,其中IL-4和IL-13能够促进B细胞分化为浆细胞,最后产生免疫球蛋白E(immunoglobulin E,IgE);IgE与肥大细胞表面受体结合后形成致敏状态,当再次暴露于变应原时,肥大细胞通过脱颗粒释放多种炎症介质,如组胺、半胱氨酸白三烯(cysteinyl leukotriene,cysLT)和前列腺素D2(prostaglandin D2,PGD2),诱导变应性气道炎症;黑色虚线右侧概括了非变应性EA的发生机制,即环境中的非变应原(PM2.5、臭氧及病原体等)刺激气道上皮细胞释放警报素,后者能够与2型固有淋巴细胞(type 2 innate lymphocytes,ILC2)相应的受体结合,激活的ILC2能够释放大量IL-5、IL-13、IL-9等炎症因子,介导气道嗜酸性粒细胞炎症发生。两种表型EA都是以IL-5作为关键炎症因子,IL-5能够促进Eos的存活及分化,并诱导Eos释放主要碱性蛋白(major basic protein,MBP)、嗜酸性粒细胞阳离子蛋白(eosinophil cationic protein,ECP)、嗜酸性粒细胞过氧化物酶(EPO)和嗜酸性粒细胞衍生神经毒素(eosinophil derived neurotoxin,EDN)、cysLT、PDG2和血小板激活因子(platelet activating factor,PAF)等炎症介质,引起气道黏液分泌增加、平滑肌细胞增生、气道高反应性(airway hyperresponsiveness,AHR)、气道重塑等病理改变;此外,Eos释放的毒性蛋白颗粒(如MBP、ECP、EPO及EDN)能形成嗜酸性粒细胞胞外陷阱(eosinophil extracellular trap,EET),而Eos发生细胞溶解时可在气道内形成夏科-雷登结晶(Charcot-Leyden crystal,CLC),以上因素进一步加重了哮喘气道炎症;TCR:T细胞受体(T cell receptor);HLA:人白细胞抗原(human leukocyte antigen)

| [1] |
GBD 2015 Chronic Respiratory Disease Collaborators Global, regional, and national deaths, prevalence, disability-adjusted life years, and years lived with disability for chronic obstructive pulmonary disease and asthma, 1990-2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet Respir Med, 2017, 5 (9): 691-706. doi: 10.1016/S2213-2600(17)30293-X.
doi: 10.1016/S2213-2600(17)30293-X URL |
| [2] |
Heaney LG, Perez de Llano L, Al-Ahmad M, et al. Eosinophilic and Noneosinophilic Asthma: An Expert Consensus Framework to Characterize Phenotypes in a Global Real-Life Severe Asthma Cohort. Chest, 2021, 160 (3): 814-830. doi: 10.1016/j.chest.2021.04.013.
doi: 10.1016/j.chest.2021.04.013 pmid: 33887242 |
| [3] |
de Groot JC, Ten Brinke A, Bel EH. Management of the patient with eosinophilic asthma: a new era begins. ERJ Open Res, 2015, 1 (1): 00024-2015. doi: 10.1183/23120541.00024-2015.
doi: 10.1183/23120541.00024-2015 |
| [4] |
Hammad H, Lambrecht BN. The basic immunology of asthma. Cell, 2021, 184(9): 2521-2522. doi: 10.1016/j.cell.2021.04.019.
doi: 10.1016/j.cell.2021.04.019 URL |
| [5] |
Kaur R, Chupp G. Phenotypes and endotypes of adult asthma: Moving toward precision medicine. J Allergy Clin Immunol, 2019, 144 (1): 1-12. doi: 10.1016/j.jaci.2019.05.031.
doi: 10.1016/j.jaci.2019.05.031 URL |
| [6] |
Van Hulst G, Bureau F, Desmet CJ. Eosinophils as Drivers of Severe Eosinophilic Asthma: Endotypes or Plasticity? Int J Mol Sci, 2021, 22 (18): 10150. doi: 10.3390/ijms221810150.
doi: 10.3390/ijms221810150 URL |
| [7] |
Lambrecht BN, Hammad H. The immunology of asthma. Nat Immunol, 2015, 16(1): 45-56. doi: 10.1038/ni.3049.
doi: 10.1038/ni.3049 pmid: 25521684 |
| [8] |
Bohrer AC, Castro E, Hu Z, et al. Eosinophils are part of the granulocyte response in tuberculosis and promote host resistance in mice. J Exp Med, 2021, 218 (10): e20210469. doi: 10.1084/jem.20210469.
doi: 10.1084/jem.20210469 |
| [9] |
Weller PF, Spencer LA. Functions of tissue-resident eosinophils. Nat Rev Immunol, 2017, 17(12): 746-760. doi: 10.1038/nri.2017.95.
doi: 10.1038/nri.2017.95 pmid: 28891557 |
| [10] |
Shah K, Ignacio A, McCoy KD, et al. The emerging roles of eosinophils in mucosal homeostasis. Mucosal Immunol, 2020, 13(4): 574-583. doi: 10.1038/s41385-020-0281-y.
doi: 10.1038/s41385-020-0281-y URL |
| [11] |
Mesnil C, Raulier S, Paulissen G, et al. Lung-resident eosinophils represent a distinct regulatory eosinophil subset. J Clin Invest, 2016, 126 (9): 3279-3295. doi: 10.1172/JCI85664.
doi: 10.1172/JCI85664 URL |
| [12] |
Van Hulst G, Batugedara HM, Jorssen J, et al. Eosinophil diversity in asthma. Biochem Pharmacol, 2020, 179: 113963. doi: 10.1016/j.bcp.2020.113963.
doi: 10.1016/j.bcp.2020.113963 URL |
| [13] |
Wechsler ME, Munitz A, Ackerman SJ, et al. Eosinophils in Health and Disease: A State-of-the-Art Review. Mayo Clin Proc, 2021, 96 (10): 2694-2707. doi: 10.1016/j.mayocp.2021.04.025.
doi: 10.1016/j.mayocp.2021.04.025 pmid: 34538424 |
| [14] |
Granger V, Zerimech F, Arab J, et al. Blood eosinophil cationic protein and eosinophil-derived neurotoxin are associated with different asthma expression and evolution in adults. Thorax, 2022, 77(6): 552-562. doi: 10.1136/thoraxjnl-2021-217343.
doi: 10.1136/thoraxjnl-2021-217343 URL |
| [15] |
Choi Y, Le Pham D, Lee DH, et al. Biological function of eosinophil extracellular traps in patients with severe eosinophilic asthma. Exp Mol Med, 2018, 50 (8): 1-8. doi: 10.1038/s12276-018-0136-8.
doi: 10.1038/s12276-018-0136-8 |
| [16] |
Ueki S, Konno Y, Takeda M, et al. Eosinophil extracellular trap cell death-derived DNA traps: Their presence in secretions and functional attributes. J Allergy Clin Immunol, 2016, 137 (1): 258-267. doi: 10.1016/j.jaci.2015.04.041.
doi: 10.1016/j.jaci.2015.04.041 URL |
| [17] |
Lu Y, Huang Y, Li J, et al. Eosinophil extracellular traps drive asthma progression through neuro-immune signals. Nat Cell Biol, 2021, 23 (10): 1060-1072. doi: 10.1038/s41556-021-00762-2.
doi: 10.1038/s41556-021-00762-2 URL |
| [18] |
Grozdanovic MM, Doyle CB, Liu L, et al. Charcot-Leyden crystal protein/galectin-10 interacts with cationic ribonucleases and is required for eosinophil granulogenesis. J Allergy Clin Immunol, 2020, 146 (2): 377-389.e10. doi: 10.1016/j.jaci.2020.01.013.
doi: S0091-6749(20)30100-7 pmid: 31982451 |
| [19] |
Persson EK, Verstraete K, Heyndrickx I, et al. Protein crystallization promotes type 2 immunity and is reversible by antibody treatment. Science, 2019, 364 (6442): eaaw4295. doi: 10.1126/science.aaw4295.
doi: 10.1126/science.aaw4295 |
| [20] |
Mack EA, Pear WS. Transcription factor and cytokine regulation of eosinophil lineage commitment. Curr Opin Hematol, 2020, 27 (1): 27-33. doi: 10.1097/MOH.0000000000000552.
doi: 10.1097/MOH.0000000000000552 URL |
| [21] |
Zustakova M, Kratochvilova L, Slama P. Apoptosis of Eosino-phil Granulocytes. Biology (Basel), 2020, 9 (12): 457. doi: 10.3390/biology9120457.
doi: 10.3390/biology9120457 |
| [22] |
Gour N, Wills-Karp M. IL-4 and IL-13 signaling in allergic airway disease. Cytokine, 2015, 75 (1): 68-78. doi: 10.1016/j.cyto.2015.05.014.
doi: 10.1016/j.cyto.2015.05.014 URL |
| [23] |
Godar M, Deswarte K, Vergote K, et al. A bispecific antibody strategy to target multiple type 2 cytokines in asthma. J Allergy Clin Immunol, 2018, 142 (4): 1185-1193. e4. doi: 10.1016/j.jaci.2018.06.002.
doi: 10.1016/j.jaci.2018.06.002 URL |
| [24] |
Gowthaman U, Chen JS, Zhang B, et al. Identification of a T follicular helper cell subset that drives anaphylactic IgE. Science, 2019, 365 (6456): eaaw6433. doi: 10.1126/science.aaw6433.
doi: 10.1126/science.aaw6433 |
| [25] |
Sugita K, Steer CA, Martinez-Gonzalez I, et al. Type 2 innate lymphoid cells disrupt bronchial epithelial barrier integrity by targeting tight junctions through IL-13 in asthmatic patients. J Allergy Clin Immunol, 2018, 141 (1): 300-310.e11. doi: 10.1016/j.jaci.2017.02.038.
doi: 10.1016/j.jaci.2017.02.038 URL |
| [26] |
Wang W, Li Y, Lv Z, et al. Bronchial Allergen Challenge of Patients with Atopic Asthma Triggers an Alarmin (IL-33, TSLP, and IL-25) Response in the Airways Epithelium and Submucosa. J Immunol, 2018, 201 (8): 2221-2231. doi: 10.4049/jimmunol.1800709.
doi: 10.4049/jimmunol.1800709 pmid: 30185520 |
| [27] |
李赞华. 白细胞介素25与33在支气管哮喘发生中的作用. 结核病与肺部健康杂志, 2017, 6 (2): 190-192. doi: 10.3969/j.issn.2095-3755.2017.02.024.
doi: 10.3969/j.issn.2095-3755.2017.02.024 |
| [28] |
Machida K, Aw M, Salter BMA, et al. The Role of the TL1A/DR3 Axis in the Activation of Group 2 Innate Lym-phoid Cells in Subjects with Eosinophilic Asthma. Am J Respir Crit Care Med, 2020, 202 (8): 1105-1114. doi: 10.1164/rccm.201909-1722OC.
doi: 10.1164/rccm.201909-1722OC URL |
| [29] |
Brusselle GG, Maes T, Bracke KR. Eosinophils in the spotlight: Eosinophilic airway inflammation in nonallergic asthma. Nat Med, 2013, 19 (8): 977-979. doi: 10.1038/nm.3300.
doi: 10.1038/nm.3300 pmid: 23921745 |
| [30] |
Brandt EB, Bolcas PE, Ruff BP, et al. IL33 contributes to diesel pollution-mediated increase in experimental asthma severity. Allergy, 2020, 75 (9): 2254-2266. doi: 10.1111/all.14181.
doi: 10.1111/all.14181 pmid: 31922608 |
| [31] |
Brandt EB, Bolcas PE, Ruff BP, et al. TSLP contributes to allergic airway inflammation induced by diesel exhaust particle exposure in an experimental model of severe asthma. Clin Exp Allergy, 2020, 50(1):121-124. doi: 10.1111/cea.13512.
doi: 10.1111/cea.13512 pmid: 31610053 |
| [32] |
Michaudel C, Mackowiak C, Maillet I, et al. Ozone exposure induces respiratory barrier biphasic injury and inflammation controlled by IL-33 J Allergy Clin Immunol, 2018, 142(3): 942-958. doi: 10.1016/j.jaci.2017.11.044.
doi: S0091-6749(18)30028-9 pmid: 29331644 |
| [33] |
Hiraishi Y, Yamaguchi S, Yoshizaki T, et al. IL-33, IL-25 and TSLP contribute to development of fungal-associated protease-induced innate-type airway inflammation. Sci Rep, 2018, 8 (1): 18052. doi: 10.1038/s41598-018-36440-x.
doi: 10.1038/s41598-018-36440-x URL |
| [34] |
Roan F, Obata-Ninomiya K, Ziegler SF. Epithelial cell-derived cytokines: more than just signaling the alarm. J Clin Invest, 2019, 129(4): 1441-1451. doi: 10.1172/JCI124606.
doi: 10.1172/JCI124606 URL |
| [35] |
Kato A. Group 2 Innate Lymphoid Cells in Airway Diseases. Chest, 2019, 156 (1): 141-149. doi: 10.1016/j.chest.2019.04.101.
doi: 10.1016/j.chest.2019.04.101 URL |
| [36] |
Lee HS, Park DE, Lee JW, et al. Role of interleukin-23 in the development of nonallergic eosinophilic inflammation in a murine model of asthma. Exp Mol Med, 2020, 52(1): 92-104. doi: 10.1038/s12276-019-0361-9.
doi: 10.1038/s12276-019-0361-9 URL |
| [37] |
Wangberg H, White AA. Aspirin-exacerbated respiratory disease. Curr Opin Immunol, 2020, 66: 9-13. doi: 10.1016/j.coi.2020.02.006.
doi: S0952-7915(20)30022-4 pmid: 32299015 |
| [38] |
Li KL, Lee AY, Abuzeid WM. Aspirin Exacerbated Respiratory Disease: Epidemiology, Pathophysiology, and ManagementMed Sci (Basel), 2019, 7(3): 45. doi: 10.3390/medsci7030045.
doi: 10.3390/medsci7030045 |
| [39] |
Reddel HK, Bacharier LB, Bateman ED, et al. Global Initiative for Asthma Strategy 2021: executive summary and rationale for key changes. Eur Respir J, 2021, 59 (1): 2102730. doi: 10.1183/13993003.02730-2021.
doi: 10.1183/13993003.02730-2021 URL |
| [40] |
Bakakos A, Loukides S, Bakakos P. Severe Eosinophilic Asthma. J Clin Med, 2019, 8(9): 1375. doi: 10.3390/jcm8091375.
doi: 10.3390/jcm8091375 URL |
| [41] |
Burgess JK, Jonker MR, Berg M, et al.Periostin: contributor to abnormal airway epithelial function in asthma? Eur Respir J, 2021, 57 (2): 2001286. doi: 10.1183/13993003.01286-2020.
doi: 10.1183/13993003.01286-2020 URL |
| [42] |
Chen M, Shepard K 2nd, Yang M, et al. Overlap of allergic, eosinophilic and type 2 inflammatory subtypes in moderate-to-severe asthma. Clin Exp Allergy, 2021, 51 (4): 546-555. doi: 10.1111/cea.13790.
doi: 10.1111/cea.13790 URL |
| [43] |
de Groot JC, Storm H, Amelink M, et al. Clinical profile of patients with adult-onset eosinophilic asthma. ERJ Open Res, 2016, 2 (2): 00100-2015. doi: 10.1183/23120541.00100-2015.
doi: 10.1183/23120541.00100-2015 |
| [44] |
Bousquet J, Humbert M, Gibson PG, et al. Real-World Effectiveness of Omalizumab in Severe Allergic Asthma: A Meta-Analysis of Observational Studies. J Allergy Clin Immunol Pract, 2021, 9 (7): 2702-2714. doi: 10.1016/j.jaip.2021.01.011.
doi: 10.1016/j.jaip.2021.01.011 URL |
| [45] |
Humbert M, Taillé C, Mala L, et al. Omalizumab effectiveness in patients with severe allergic asthma according to blood eosinophil count: the STELLAIR study. Eur Respir J, 2018, 51 (5):1702523. doi: 10.1183/13993003.02523-2017.
doi: 10.1183/13993003.02523-2017 |
| [46] |
Busse WW, Bleecker ER, FitzGerald JM, et al. Long-term safety and efficacy of benralizumab in patients with severe, uncontrolled asthma: 1-year results from the BORA phase 3 extension trial. Lancet Respir Med, 2019, 7 (1): 46-59. doi: 10.1016/S2213-2600(18)30406-5.
doi: 10.1016/S2213-2600(18)30406-5 URL |
| [47] |
Agache I, Song Y, Rocha C, et al. Efficacy and safety of treatment with dupilumab for severe asthma: A systematic review of the EAACI guidelines-Recommendations on the use of biologicals in severe asthma. Allergy, 2020, 75 (5): 1058-1068. doi: 10.1111/all.14268.
doi: 10.1111/all.14268 pmid: 32154939 |
| [48] |
Agache I, Beltran J, Akdis C, et al. Efficacy and safety of treatment with biologicals (benralizumab, dupilumab, mepolizumab, omalizumab and reslizumab) for severe eosinophilic asthma. A systematic review for the EAACI Guidelines-recommendations on the use of biologicals in severe asthma. Allergy, 2020, 75 (5): 1023-1042. doi: 10.1111/all.14221.
doi: 10.1111/all.14221 pmid: 32034960 |
| [49] |
Corren J, Parnes JR, Wang L, et al. Tezepelumab in Adults with Uncontrolled Asthma. N Engl J Med, 2017, 377 (10): 936-946. doi: 10.1056/NEJMoa1704064.
doi: 10.1056/NEJMoa1704064 URL |
| [50] |
Menzies-Gow A, Corren J, Bourdin A, et al. Tezepelumab in Adults and Adolescents with Severe, Uncontrolled Asthma. N Engl J Med, 2021, 384 (19): 1800-1809. doi: 10.1056/NEJMoa2034975.
doi: 10.1056/NEJMoa2034975 URL |
| [1] | 何燕超, 揭志军. 综合医院活动性肺结核的早期筛查策略[J]. 结核与肺部疾病杂志, 2022, 3(4): 334-337. |
| [2] | 周伊南, 朱惠莉. 慢性阻塞性肺疾病合并肺结核的研究进展[J]. 结核与肺部疾病杂志, 2022, 3(4): 338-342. |
| [3] | 张彦坤, 关艳, 韩朝, 章志华. 18例结核性后葡萄膜炎患者临床特征分析[J]. 结核与肺部疾病杂志, 2022, 3(3): 216-221. |
| [4] | 韩玮欣, 赵立明, 程珊珊, 孟艺哲, 陈颜强. 15例中枢神经系统结核合并脑动脉内膜炎患者的临床特征[J]. 结核与肺部疾病杂志, 2022, 3(3): 227-230. |
| [5] | 郭华征, 邢振川. 北京市某综合医院呼吸科病房肺结核患者特征及转诊情况分析[J]. 结核与肺部疾病杂志, 2022, 3(2): 110-117. |
| [6] | 朱丹, 陈燕, 双庆翠, 曾慧卉. 慢性阻塞性肺疾病急性加重期与慢性阻塞性肺疾病合并社区获得性肺炎的临床观察性分析[J]. 结核与肺部疾病杂志, 2022, 3(2): 118-124. |
| [7] | 张思琪, 李静, 仵倩红. 结核分枝杆菌扫描系统在结核病诊断中的应用价值[J]. 结核与肺部疾病杂志, 2022, 3(2): 170-172. |
| [8] | 刘辉敏, 田瑶, 贝承丽, 傅满姣. 活动性肺结核免疫学检测技术应用现状及展望[J]. 结核与肺部疾病杂志, 2022, 3(1): 70-74. |
| [9] | 林慧敏, 谢佳星. 重症哮喘应用贝那利珠单抗治疗二例[J]. 结核与肺部疾病杂志, 2022, 3(1): 80-84. |
| [10] | 周琳, 沈兴华. 皮肤结核一例[J]. 结核与肺部疾病杂志, 2022, 3(1): 85-86. |
| [11] | 魏建华, 郭涛, 高小娜, 伍魏, 郭蕾. 151例住院学生结核病患者临床特征和治疗转归情况[J]. 结核与肺部疾病杂志, 2021, 2(4): 330-335. |
| [12] | 黄林欢, 周厚仕, 林琬, 林麒. 非结核分枝杆菌病17例临床分析[J]. 结核与肺部疾病杂志, 2021, 2(4): 352-354. |
| [13] | 吴迪, 范欣欣, 沈建山, 林友飞, 陈晓红, 黄明翔, 陈力舟. 联合检测凝血筛查指标及D-二聚体对新型冠状病毒肺炎患者临床分型的诊断价值[J]. 结核与肺部疾病杂志, 2021, 2(4): 355-360. |
| [14] | 中国防痨协会. 非活动性肺结核诊断及预防发病专家共识[J]. 结核与肺部疾病杂志, 2021, 2(3): 197-201. |
| [15] | 张群成, 李向南, 徐志伟, 孙冠男, 杨会珍, 程东军, 张晓菊. 经电子气管镜冷冻肺活检在肺外周结节诊断中的价值[J]. 结核与肺部疾病杂志, 2021, 2(3): 205-209. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
 京公安网备11010202008787号
ip访问总数: ip当日访问总数: 当前在线人数:
京公安网备11010202008787号
ip访问总数: ip当日访问总数: 当前在线人数:
 本作品遵循Creative Commons Attribution 3.0 License授权许可
本作品遵循Creative Commons Attribution 3.0 License授权许可